 As a practical consequence of this mixing of diatomic molecules, the wavefunctions s+s and p z+p z molecular orbitals become blended. Quantum theory also indicates that molecular orbitals (MO) of identical symmetry actually mix or hybridize. By this definition, common forms of sigma bonds are s+s, p z+p z, s+p z and d z 2+d z 2 (where z is defined as the axis of the bond or the internuclear axis). In this formal approach, a σ-bond is symmetrical with respect to rotation about the bond axis. Sigma bonding is most simply defined for diatomic molecules using the language and tools of symmetry groups. They are formed by head-on overlapping between atomic orbitals. 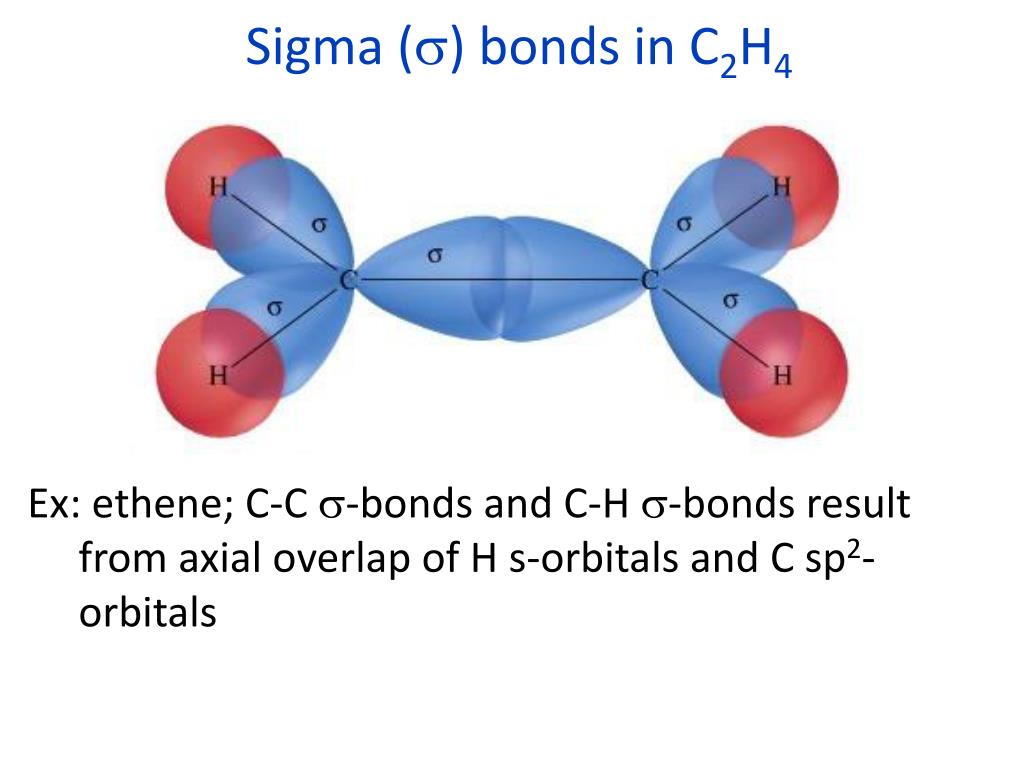
In chemistry, sigma bonds ( σ bonds) are the strongest type of covalent chemical bond. Posts will be automatically removed once it reaches a certain threshold of reports or it will be removed earlier if there is sufficient reports for manual takedown trigger.Covalent chemical bond σ bond between two atoms: localization of electron density If there is a rule violation, inform the OP and report the offending content. OPs can lock their thread by commenting /lockĤ. Comments containing case-insensitive **Answer:** or **Hence** will automatically re-flair post to ✔ Answered non-top level comments containing case-insensitive **Therefore** or **Thus** will automatically re-flair to -Pending OP Replyģ. Only downvote questions that do not abide by our rules or was asked in bad faith, NOT because the question is easy.Ģ. Upvote questions that you recognise but you cannot do. How was your experience in this subreddit? Let us know how can we do better by taking part in our survey here. by stating the syllabus requirements or presenting incorrect working/thought process towards the question.Post must contain instructor prompt or or a failed attempt of the question.Post title must be structured to classify the question properly.If this post violates our subreddit rules, please report it and feel free to manually trigger a takedown. Pastebin / / IDEOne Revert to older template If you have trouble with spacing, add some blank spaces with įor more commands, please see User Moderation Please contact u/Oryv if the bot malfunctions, or if you have questions on how to use it. Simply mention the bot in a comment and put the expressions that need to be rendered in code blocks, and u/LaTeX4Reddit will respond. To use LaTeX on this subreddit, use u/LaTeX4Reddit. Accept answers at your own risk.įor citation questions, check the Purdue Online Writing Lab Keep in mind that we do not and will not have any sort of vetting procedure for responders. This includes asking for "likes," page views, or similar things. 
Offers or solicitations of payment in any form. Requests for help with cheating, plagiarism, or other violations of academic integrity violations of copyright or terms of use or other illegal or unethical activities. 
Posts tagged "urgent," "ASAP," "important," "due in an hour," etc. "Do this for me" posts such as posting of quizzes or lists of questions. When your question has been answered, please edit the post's flair to " ✓ Answered".Ĭopied questions without context or explanation. What does your instructor want you to accomplish? Still acceptable…, but preferably not: Grade 1-6 (Pri) Grade 7-10 (Sec) Grade 11-12 (Pre-Uni) University/College ❗️ READ THE RULES BEFORE POSTING 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
